Host Cell Protein (HCP) Analysis
Host cell proteins (HCPs) are low-level, process-related protein impurities in drug products derived from the host organism in samples from process development or manufacture. Even at low level, host cell proteins (HCPs) accompanied with recombinant biotherapeutics can significantly affect stability of drug product and cause risks for immunogenicity. Detection and quantification of residual HCPs as potential process-related impurities is critical for biopharmaceutical companies in accordance with regulatory agency guidelines.
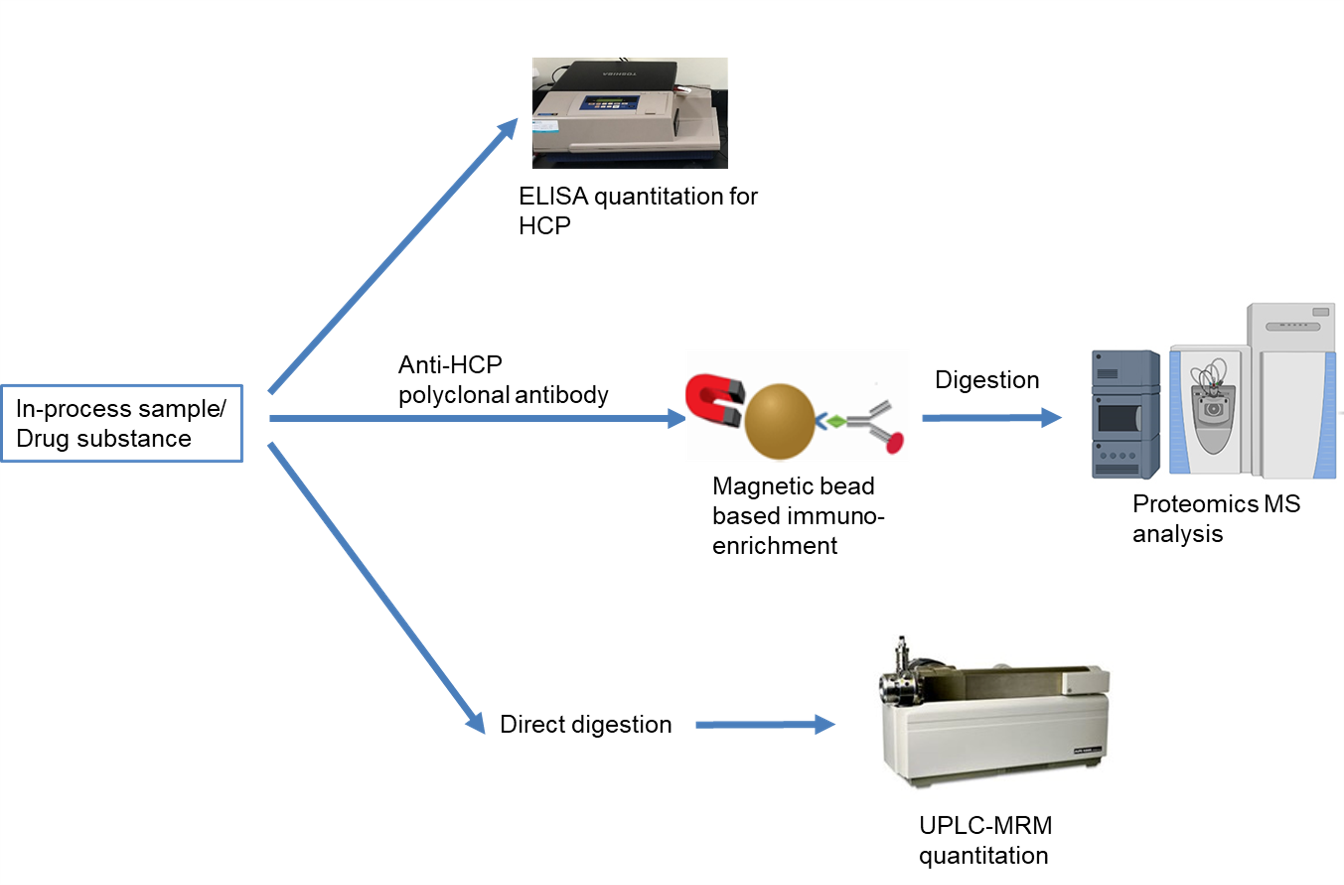
At Novabioassays, we provide three types of services for HCP analysis. The first type of service is ELISA based quantitation of HCPs with either commercially available ELISA reagents or client provided anti-HCP polyclonal antibodies. The second type of service is proteomics characterization of HCPs in process development or manufacture samples. The HCPs can be enriched by various methods, digested, and then characterized by nanoLC coupled with high resolution mass spectrometry. The third type of service is UPLC-MRM-MS based targeted quantitation of a specific HCP such as lipase PLBD2.